
Dr. Catherine Hong Huan HOR

Contact Information
Tel: 3411 5600
Email: catherinehor@hkbu.edu.hk
Website: https://chem.hkbu.edu.hk/hor
Assistant Professor, Department of Chemistry
- SYSTEM HEALTH
Dr. Hor’s first scientific training began as a research assistant at the National University of Singapore, working in a molecular neurobiology lab under the guidance of Professor Bor Luen TANG. She then pursued her PhD majoring in developmental genetics at The University of Hong Kong, studying mouse hindbrain neural tube patterning and Hedgehog signaling under the supervision of Professor Mai Har SHAM and Professor Chi Chung HUI at the Department of Biochemistry. Upon graduation, she joined Dr. Eyleen Goh LK's lab at Duke-NUS Medical School Singapore as a postdoctoral research fellow, pursuing research topics in the realm of developmental and regenerative neuroscience.
In 2018, Dr. Hor obtained her first independent research grant, which is a prestigious and competitive national level research fund for young investigator-Young Individual Research Grand (YIRG) funded by the Ministry of Health, Singapore. In 2019, Catherine joined HKBU and established her lab at HKBU as a faculty member of the Department of Chemistry. In 2020, Dr. Hor was awarded one of the most competitive major research grant in Hong Kong, Collaborative Research Fund (CRF), in which she leads a team of international scientists from Hong Kong, Singapore and mainland China to investigate the cellular and neurobiology of viral infection.
Her research strives to understand the molecular and cellular pathology of hereditary neurological and neuropsychiatric disorders, with particular interest in deciphering the neurochemistry and neurological roles of an under-appreciated but instrumental cell-cell signaling organelle, the primary cilium. Her current projects involve mouse disease models, and human induced-pluripotent stem cells disease modeling of ciliopathy-like disorders. Her cross-disciplinary collaborative projects with local and international scientists focus on the development of non-invasive biomarkers and therapeutics targeting pediatric neuropsychiatric disorders.
PROJECT HIGHLIGHTS:
The Biochemistry of Primary Cilium in Neurodevelopment
The primary cilium is a non-motile cilium found on the surface of nearly every cell types. It functions primarily as an “antenna’’ on the cell membrane to capture and transduce signals from extracellular signaling molecules to the cytoplasm and nucleus. We aim to decipher the molecular machineries underlying the biogenesis and signal transduction in the primary cilium, particularly, in the context of neurochemistry and neurobiology.
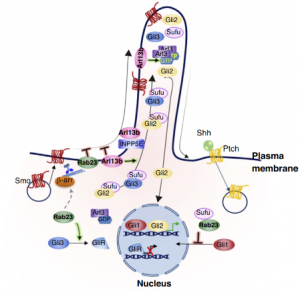
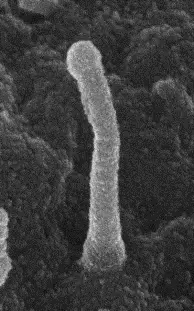
.png)
Scanning electron microscopy image (Left) and Transmission electron microscopy image (Right) of a primary cilium in the brain. Hor CHH & Goh EL. Curr Opin Genet Dev. 2019, 56, 61-68.
Human Stem Cell & Disease Modeling of Ciliopathy and Neurological Disorders
Impairment of the primary cilia adversely affects the physiological functions of multiple organs including the brain. In children, defective primary cilia lead to a class of heritable disorders collectively known as ciliopathies. We use CRISPR/Cas9 genome editing and induced-pluripotent stem cells (iPSC) technologies to establish the disease models of congenital diseases such as ciliopathies in animal and cellular platforms. Using these disease models in combination with biochemistry assays, comparative proteomics and genomics approaches, we aim to study the disease mechanism of ciliopathies; as well as to develop new therapeutics targeting ciliopathies.

(Left) Immunohistology images show primary cilia (green) in the Purkinje layer of the adult mouse cerebellum. Red: Purkinje cells. (Right) Genetically modified embryonic mouse model of ciliopathy. Hor CH & Goh EL. Cerebral Cortex. 2018, 28(4), 1516-1531. Hor CH* & Goh EL*. Rev in the Neurosciences 2018, 29(8), 849-860.
Neurobiology of viral infection
Emerging clinical observations show that SARS-Cov-2 infected patients developed various neurological manifestations. Using humanized-mouse model, human stem cells, and brain organoid as the research platforms, we decipher the cellular and subcellular basis underlying SARS-Cov-2 infection in the central nervous system.
MEDIA COVERAGE:
【抗疫科研女將】研細胞「小天線」 探新冠損腦之謎
View More: News



